Measuring the molecular and chemical composition of biological samples remains challenging. Yet, medical advances depend on detecting and identifying molecular disease signatures. They also rely on developing treatments that target specific illnesses within individuals. Biological samples are fragile by nature. Therefore, prolonged exposure to spectroscopic investigation can damage them. Moreover, many biological materials auto-fluoresce, which interferes with the acquisition of clear Raman spectra. At ultraviolet wavelengths, fluorescence and Raman signals become spectrally separated. Consequently, Deep UV Raman spectroscopy enables the detection, identification, and quantification of substances at much lower concentrations. This capability surpasses near-UV, visible, or infrared (IR) methods.
Odin™, IS-Instruments’ compact Deep UV Raman spectrometer, operates at 228.5 nm. It integrates the cooled CCD detector from the HES spectrometer. As a result, Odin™ measures biological samples more effectively than previous systems without degrading fragile specimens (Foster et al., 2022). Researchers now propose that this technique could facilitate direct detection of disease in tissues.
Collaboration with the Royal Veterinary College Surgeon
IS-Instruments has engaged in ongoing discussions with Dr Jay Dudhia at the Royal Veterinary College. He previously applied Raman spectroscopy to joint tissues (see Gaifulina et al Clinical Spectroscopy 3 2021 100012). Following a briefing on Odin™, ISI, and Dr Dudhia conducted a pilot study using equine articular joint synovial fluid samples.
Synovial fluid is a non-Newtonian substance found within joint cavities. It contains proteins derived from blood plasma and from cells within joint tissues. A detailed analysis of this fluid could enable the early detection of diseases such as arthritis. Raman spectroscopy may provide a path toward that capability.
Overcoming Fluorescence in Complex Biologics
Like many biological materials, synovial fluid exhibits strong fluorescence, which masks the Raman signal. Consequently, applying Raman spectroscopy to such samples has proven to be challenging. Nevertheless, researchers have obtained Raman spectra from synovial fluid using surface-enhanced Raman spectroscopy (SERS). In SERS, gold or silver nanoparticles are added to generate a resonant effect that amplifies the Raman signal intensity. However, this process requires additional preparation and can alter the sample. Deep UV Raman spectroscopy with Odin™ eliminates that need. It enables clear spectral acquisition without the need for additives or complex sample conditioning. Therefore, Odin™ provides a more straightforward, non-destructive approach for analysing biological materials.
Results from Equine Synovial Fluid Analysis
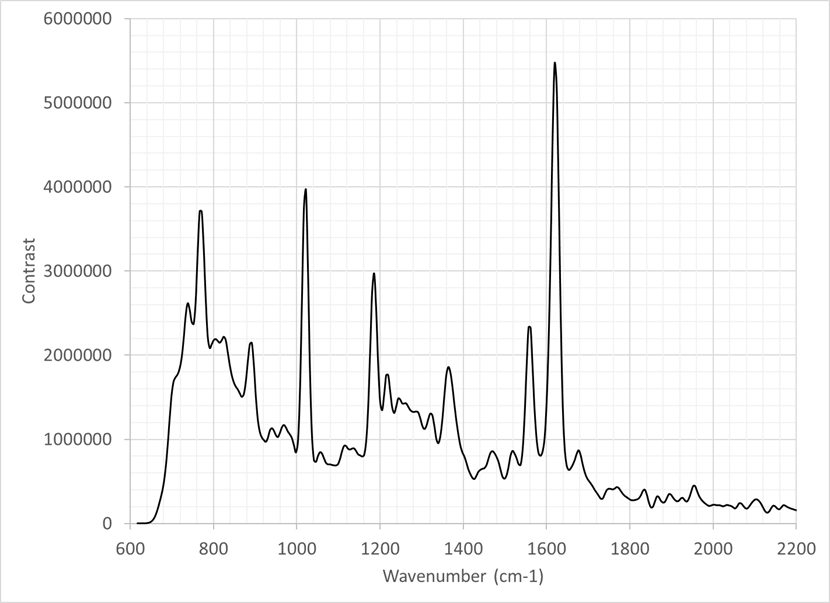
The figure shows the Raman spectrum from an equine synovial fluid sample measured using Odin™. The data was collected with a 30-second integration time and averaged over ten frames. Only minimal processing was applied, including the removal of cosmic rays. The resulting power spectrum displays discrete peaks from 750 cm⁻¹ to 1700 cm⁻¹. Several peaks correspond to known functional groups and amino acids.
This confirms that Odin™ can analyse biological and biochemical materials non-destructively. In addition, it achieves this without the sample preparation required by other techniques, including SERS.
Conclusion
Odin™ demonstrates the ability of deep UV Raman spectroscopy to analyse delicate biological samples with exceptional sensitivity. Moreover, it reduces interference from fluorescence while preserving the sample’s integrity. As a result, it represents a significant step forward in non-destructive biomedical spectroscopy and the early detection of disease.